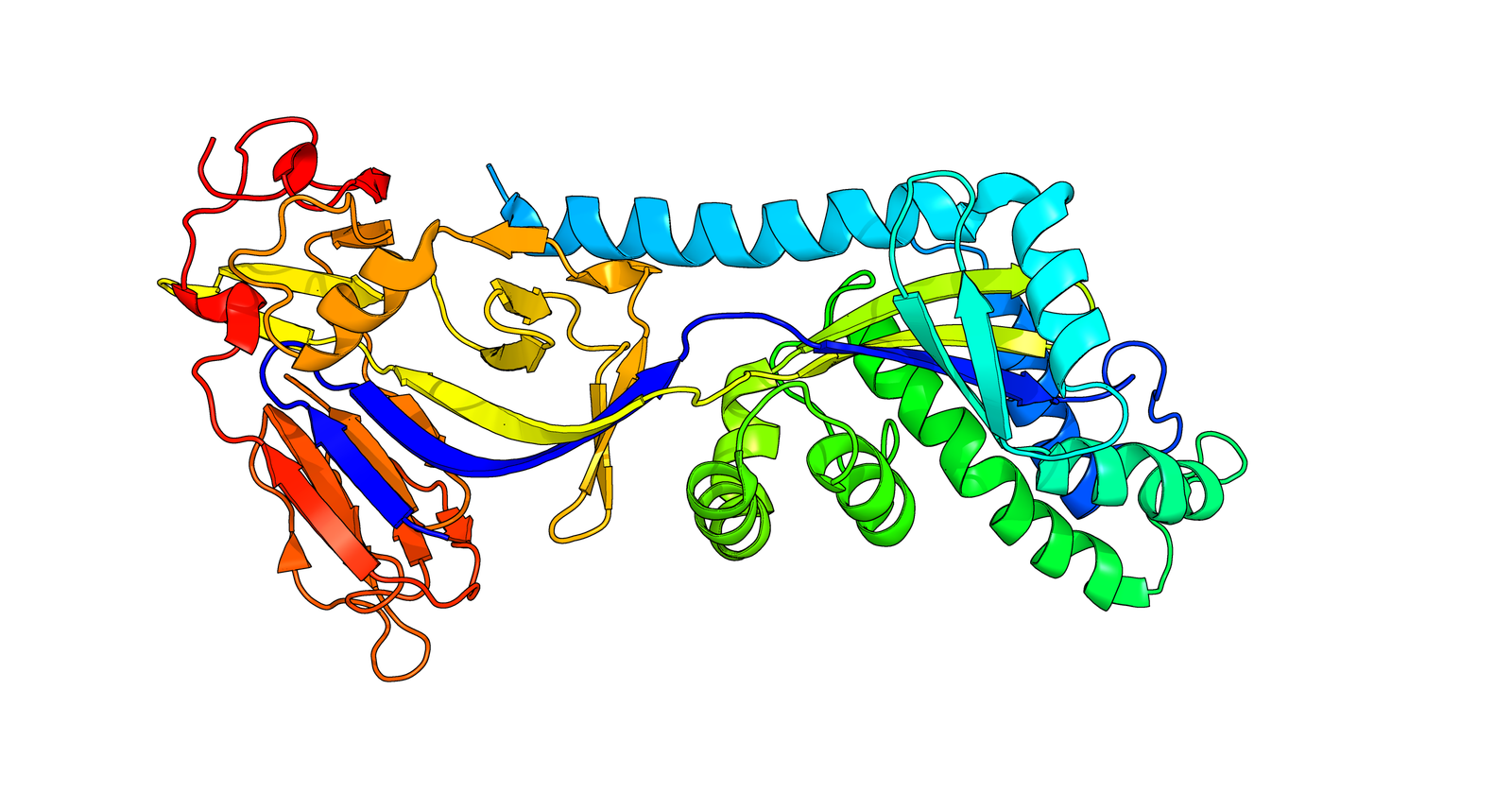
Structure of Fusion glycoprotein from HMPV
3D Structure
| Method | PDB ID | Downloads |
|---|---|---|
| X-ray | --- | Click here |
| Cryo-EM | 7UR4 | Click here |
| X-ray | 8VT3 | Click here |
Functional Information
Fusion glycoprotein F0, inactive precursor that is cleaved to give rise to the mature F1 and F2 fusion glycoproteins while Fusion glycoprotein F1 which is a class I viral fusion protein has at least 3 conformational states: pre-fusion native state, pre-hairpin intermediate state, and post-fusion hairpin state. During viral and plasma cell membrane fusion, the coiled coil regions assume a trimer-of-hairpins structure, positioning the fusion peptide in close proximity to the C-terminal region of the ectodomain. The formation of this structure appears to drive apposition and subsequent fusion of viral and cellular membranes leading to delivery of the nucleocapsid into the cytoplasm. This fusion is pH independent and occurs at the plasma or endosomal membrane. The trimer of F1-F2 (F protein) also facilitates the attachment to host cell by binding to host heparan sulfate. Moreover, the fusion glycoprotein F2 is a major determinant of the species specificity of RSV infection. The trimer of F1-F2 (F protein) also facilitates the attachment to host cell by binding to host heparan sulfate (PubMed:22238303).
Interactions
Fusion glycoprotein F1- Homotrimer. Heterodimer with fusion protein F2; disulfide-linked. As a heterodimer with F2, interacts with host heparan sulfate (PubMed:22238303).
- As a heterodimer with F2, interacts with host integrin ITGAV/ITGB1 (PubMed:19164533).
- Part of a complex composed of F1, F2 and G glycoproteins (By similarity).
- Homotrimer. Heterodimer with fusion protein F1; disulfide-linked. As a heterodimer with F1, interacts with host heparan sulfate (PubMed:22238303).
- As a heterodimer with F2, interacts with host integrin ITGAV/ITGB1 (PubMed:19164533).
- Part of a complex composed of F1, F2 and G glycoproteins.
Domains Orgainzation
- 1-30 Intravirion
- 31-51 Helical
- 58-219 virion surface (disordered)
Post-Translational Modification
N-linked glycosylation at Asn57, Asn172 and Asn353. The F glycoprotein is synthesized as a F0 inactive precursor that is heavily N-glycosylated and processed.